Rabipur Injection
Rabipur Injection Specification
- Dosage Form
- Injection
- Origin
- Biological/Vaccine
- Application
- Human use Immunization against rabies
- Physical Color/Texture
- Clear or slightly opalescent, colorless solution
- Fermentation Smell
- Odorless
- Enzymatic Activity
- None
- Storage Instructions
- Store at 2C to 8C; do not freeze
- Shelf Life
- Usually 24 months if properly stored
- Brand Name
- Rabipur
- Administration Route
- Intramuscular (IM), usually in the deltoid region
- Container Material
- Glass vial
- Barcode
- Present on packaging for pharmacy use
- Quality Certifications
- WHO prequalified, GMP compliant
- Contraindications
- History of severe allergic reaction to previous dose or any component
- Pack Size
- 1 single dose vial + 1 ampoule of sterile water for injection (diluent)
- Reconstitution Required
- Yes, must be reconstituted with provided sterile diluent before use
- Prescription Status
- Prescription required
About Rabipur Injection
Rabipur :Rabipur is a Powder and solvent for solution for injection Rabies vaccine, inactivated,
Pharmaceutical Form :
Powder and solvent for solution for injection. A clear colorless solution results after reconstitution of the white freeze-dried powder with the clear and colorless solvent.
Clinical Particulars:
(Therapeutic indications) Pre-exposure prophylaxis (before possible risk of exposure to rabies)b) Post-exposure prophylaxis (after known or possible exposure to rabies)Consideration should be given to national and/or WHO guidance regarding the prevention of rabies.
Posology and method of administration Posology. The recommended single intramuscular dose is 1 ml in all age groups. Whenever possible according to vaccine availability, it is recommended that one type of cell culture vaccine should be used throughout the course of pre- or post-exposure immunisation. However, adherence to the recommended schedules is of critical importance for post-exposure prophylaxis, even if another type of cell culture vaccine has to be used.
What RABIPUR looks like ?
Each pack of RABIPUR contains, one vial of vaccine powder, one ampoule of sterile water for injections, one disposable syringe.
Your doctor will inject the sterile water into the vial to make the liquid for your injection. This liquid is clear and colorless. Ingredients Vaccine Active ingredients: Not less than 2.5 International Units of inactivated Rabies virus (Flury LEP strain)
Other ingredients:
Trometamol Sodium chloride Disodium edetate Mono potassium glutamate Polygeline Sucrose Possible trace amounts: neomycin, chlorte tracycline,amphotericin BDiluent Water for Injections The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovinespongi form encephalitis) has resulted from the administration of any vaccine product. Contains no antimicrobial agent. Product is for single use in one patient only. Discard any residue.
Manufactured by : Novartis Vaccines and Diagnostics GmbH
Key Features of Rabipur Injection
Rabipur Injection is recognized for its quality, being both WHO prequalified and GMP compliant. It comes in a single dose glass vial that must be reconstituted with the provided sterile water before administration. As a biological vaccine for preventing rabies in humans, it is prescription-only and suited to medical settings. The packaging includes a barcode to ensure traceability and authenticity for pharmacies and distributors.
Proper Administration and Storage Guidelines
Rabipur must be reconstituted by mixing the contents of the vaccine vial and the ampoule of sterile water immediately prior to use. It is administered intramuscularly, typically into the deltoid region. The vaccine needs to be stored at a temperature between 2C and 8C and should not be frozen. Adhering to these guidelines maintains the vaccines effectiveness and shelf life, which is up to 24 months if properly stored.
FAQs of Rabipur Injection:
Q: How should Rabipur Injection be prepared before administration?
A: Rabipur Injection requires reconstitution. To prepare, mix the entire contents of the single dose glass vial with the sterile water provided in the ampoule immediately before use. Shake gently to ensure the solution is clear and ready for intramuscular injection.Q: What is the recommended route and site for administering Rabipur Injection?
A: Rabipur Injection is administered intramuscularly, most commonly in the deltoid muscle of the upper arm for adults and children. This ensures optimal absorption and efficacy of the vaccine.Q: When is Rabipur Injection indicated for use?
A: Rabipur Injection is used for the prevention of rabies following potential exposure (post-exposure prophylaxis) or as a preventive measure (pre-exposure prophylaxis), particularly for individuals at risk such as laboratory workers, veterinarians, and travelers to high-risk areas.Q: What are the storage requirements for Rabipur Injection?
A: Store Rabipur Injection at a temperature between 2C and 8C in a refrigerator. Do not freeze the vaccine, as this can compromise its effectiveness. Proper storage ensures a shelf life of up to 24 months from the date of manufacture.Q: What are the contraindications for using Rabipur Injection?
A: Rabipur Injection should not be administered to individuals with a known history of severe allergic reaction to a previous dose or any component of the vaccine. Always consult a healthcare professional before vaccination.Q: What are the key benefits of Rabipur Injection?
A: Rabipur Injection offers effective immunization against rabies, a potentially fatal viral infection. It is certified by WHO and manufactured according to GMP guidelines, providing assured quality and safety in human use.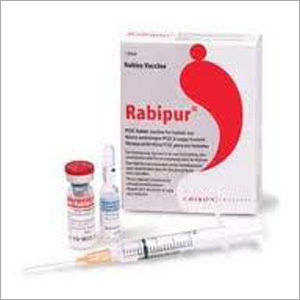

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Oncology Drugs Category
Thymocyte Globulin Injection
Price 80 INR / Piece
Minimum Order Quantity : as per requirement Piece
Storage Instructions : Store at 2C to 8C. Do not freeze. Protect from light.
Application : Other, Intravenous use in hospital settings
Salt Composition : Antithymocyte Globulin (Rabbit) 25 mg/mL
Shelf Life : 2 years from date of manufacture
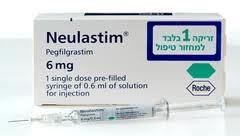
Neulastim Injection
Storage Instructions : Store refrigerated at 2C to 8C. Do not freeze.
Application : Other, Oncology supportive care
Salt Composition : Pegfilgrastim 6 mg/ml
Shelf Life : 24 months
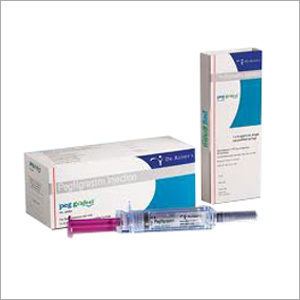
PEG Grafeel Injection
Storage Instructions : Store in a refrigerator (2C 8C); do not freeze
Application : Other, To boost white blood cell production and reduce infection risk during chemotherapy
Salt Composition : Pegfilgrastim (Filgrastim Pegylated)
Shelf Life : 24 months from date of manufacture
Life Saving Drug
Storage Instructions : Cool Place
Application : Other
Salt Composition : Aspirin 300mg
Shelf Life : 24 Months
&
We are accepting bulk order quantity.